Shedding Part 2- The Bio-Distribution and Excretion of Covid mRNA Vaccine Components and Products
In order to transmit or "shed" the Covid mRNA encapsulated lipid nanoparticle and/or the transcribed spike protein product, dissemination to organs and body fluids must occur. Does it? Answer: Yes.
OK, so in Part 1 of this series on Covid mRNA vaccine shedding, I provided evidence that:
The FDA and the EMA define the mRNA vaccines as gene therapies.
The FDA requires that gene therapy products undergo human shedding studies given the known risks of shedding
Shedding studies were not done because, even though the vaccines are gene-therapies, they legally fell under the legal definition of a “countermeasure” in a public health emergency. Countermeasures do not require shedding or other types of safety studies before mass use.
All we know is that from a FOIA obtained document, Pfizer did a shedding study on rats but we don’t know what they found.
HOW DOES SHEDDING OCCUR AND IS THERE ANY EVIDENCE OF IT OCCURRING WITH THE COVID mRNA VACCINES?
In order for the vaccine or spike to be shed, it would first require distribution of the vaccine components or spike protein product to the lungs (to then be exhaled) and other body fluids (to then be excreted)
To explore this possibility, it is important that we define what a lipid nanoparticle LNP) is, along with their natural, biological counterparts which are called exosomes or extracellular vesicles (EVs).
The papers I reviewed used the terms exosomes, LNP’s, EV’s, and even nanoparticles somewhat interchangeably although there are some differences. For instance, exosomes are a subset of extracellular vesicles (EV’s). From this paper in Molecular Therapy, they state:
Exosome-like nanovesicles (ELNVs) are biological nanostructures of 40–150 nm, are secreted by most types of cells and relay information between cells and organisms across all three kingdoms of life.1,2 Although earlier perceived to be cellular debris and hence undervalued, ELNVs are now acknowledged as crucial entities to regulate physiological functions of multicellular organisms in an intercellular transmission manner.
From another paper in Science:
Exosomes are EVs with a size range of ~40 to 160 nm (average ~100 nm) in diameter with an endosomal origin. For instance, the LNP’s in the Covid MRNA vaccines are approximately 100 - 400 nm in size.
The most important fact to remember is that the smaller the size, the more widely they distribute and the more easily they can enter the body (more on the latter later).
For context, the length of the SARS-Cov2 virus is about 9-12 nm in size. Further, as Banoun points out in her masterful review of the topic of shedding:
“Huge amounts of mRNA are injected compared to the circulation of a virus during a natural infection: up to 10 to 7 times more, according to Professor Jean-Michel Claverie [27].”
Further, there are different biologic materials that can be used to make the outer membrane enclosing the contents of a nanoparticle. Lipids (i.e. liposomes or LNP’s) are one of the most commonly used for drug delivery. Early conventional “liposomes” (yet another term) had limitations such as short half-life and rapid systemic clearance following their clearance by the reticuloendothelial system (RES). However, the conjugation of polymers such as polyethylene glycol (PEG) resulted in the generation of sterically stabilized liposomes with prolonged half-life and increased stability.
To wit, the Covid mRNA vaccines used PEG to stabilize the LNP carrying the modified mRNA.
So what are nanoparticles/LNPs/exosomes, what is inside them, and what do they do?
Basically, they are tiny sacs enclosed by a lipid membrane which can contain any of the following: proteins, metabolites, enzymes, growth factors, and nucleic acids. You can also package drugs (and synthetic mRNA) into them in order to deliver their contents into recipient cells to effectively alter their biological response.
Natural, endogenous exosomes are associated with immune responses, viral pathogenicity, pregnancy, cardiovascular diseases, central nervous system–related diseases, and cancer progression. Such exosome-mediated responses can be disease promoting or restraining. Exosomes can be engineered to deliver diverse therapeutic payloads, including short interfering RNAs, antisense oligonucleotides, chemotherapeutic agents, and immune modulators, with an ability to direct their delivery to a desired target.
Importantly, synthetic mRNA vaccine LNPs have the same structure as the natural exosomes they seek to mimic.
So what do we know about human biodistribution of synthetic LNP’s??
From this article by Sonia Elijah, regulators knew LNP’s distribute widely in the human body:
In the recent leaked letter by the EMA, Executive Director, Emer Cooke, to the Chair of COVID-19 Special Committee, MEP Kathleen Van Brempt, Cooke begrudgingly admitted, “that the lipid nanoparticles can distribute rather non-specifically to several organs such as liver, spleen, heart, kidney, lung and brain, with the liver appearing to be the organ where the lipid nanoparticles distribute most.”
Her admission was made on the heels of the Therapeutics Goods Administrations (TGA) of Australia’s evaluation report on Pfizer’s nonclinical biodistribution study, which alarmingly revealed that the lipid nanoparticles which encase the mRNA, travel to the liver, spleen, brain, eyes, bone marrow, adrenal glands, ovaries and testes– nearly every organ tissue.
Further, beyond the mRNA encased in synthetic LNP’s, “naked” mRNA as well as mRNA encased in natural LNP’s (called exosomes) and spike protein in free form or encapsulated in exosomes can be found in the bloodstream and breast milk.
More worryingly, LNP’s or their natural equivalent, exosomes (a.k.a. extracellular vesicles (EVs)) are able to be excreted through body fluids (sweat, sputum, breast milk) and to pass the transplacental barrier. These exosomes are also able to penetrate by inhalation and through healthy or injured skin as well as orally through breast milk.
This figure from a review paper on nanoparticle therapies is illuminating:
If that isn’t concerning enough, it gets worse.
The above paper from 2017 states:
Due to their unique characteristics, nanoparticles are widely used in biomedical and industrial applications (Lee et al., 2007; Zhang et al., 2008; Das et al., 2009; Vance et al., 2015). Currently, there are 1,814 marketed consumer products containing nanoparticles, including antibiotics, food items, textiles, sports tools, and electronic materials, and the number is increasing steadily.
Good to know (and unsurprising) that as of 2017, the human race has 1,814 consumer products out there relying on the use of nanoparticles and we have no comprehensive understanding of what the short or long term risks of both the absorption into our bodies and or the risk of secondarily shedding the nanoparticles onto other humans, in particular our families, friends, and others.
It is shocking that in a number of review papers on nanoparticle technology, in each one, statements like this appear: “it is clear that more studies are needed to determine the mechanisms by which NPs affect particular organ systems.”
Equally prevalent are statements such as the below from this review paper:
Despite the potential for clinical application, some studies have suggested that NPs can be toxic. These studies have demonstrated the ability of NPs to accumulate in cells and induce organ-specific toxicity. These studies, combined with the ever-increasing human exposure, demonstrate an urgent need for the design of safe NPs and the development of strict guidelines for their development with regards to toxicity testing.
Urgent need to design safe nanoparticles? Strict guidelines needed for development in regards to testing for toxicity? Bit late for that now given “we” injected transmissable nanoparticles into the bodies of billions of people of all ages across the world.
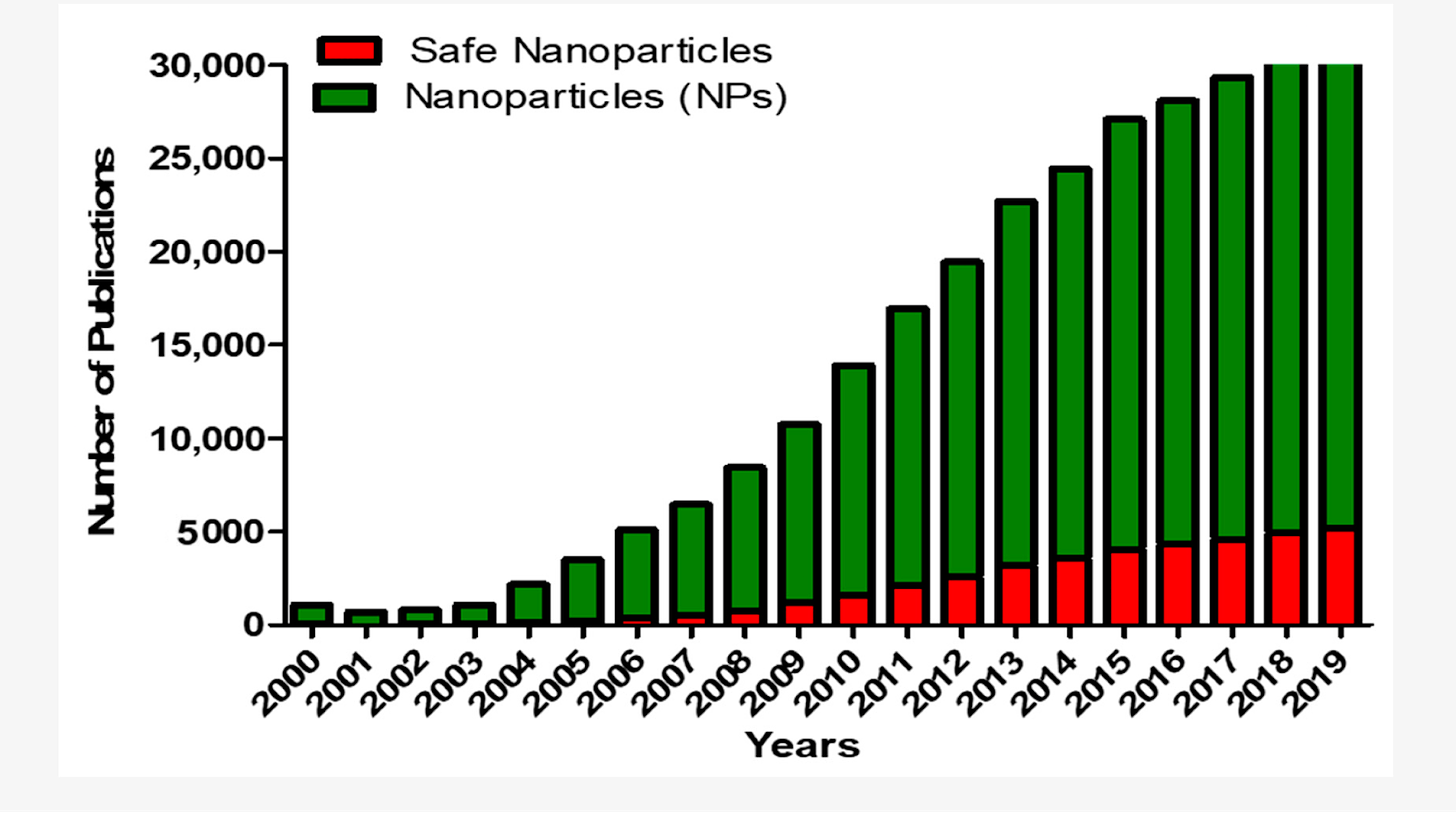
The reason for so many papers on nanoparticle technology constantly calling out for safety and shedding studies is that the researchers all are fully aware that there is a distinct paucity of studies confirming safety and/or risks of nanoparticles/LNP’s - the red bars in the chart below refers to the number of studies on nanoparticles found with titles containing the words “risks, safety or toxicity”
I find it highly troubling that the nanoparticle technology industry is expanding so rapidly yet the potential for biologic toxicity to others has been so little studied. Almost all the studies have been in animals and they are not reassuring. At all.
Ed: Sorry to interrupt but if you like (and enjoy learning from) this Substack, please consider supporting with a paid subscription (my time is under increasing strain and I need to prioritize - support will help me do that!) Thanks.
Before we get into the studies demonstrating the passing of spike protein and/or mRNA from vaccinated to the unvaccinated via various routes, know that Pfizer knew that shedding was a possibility given that they specifically excluded people “exposed” to the vaccine via inhalation (not subtle) or skin contact:
Starting on p. 67 of the protocol the investigator is instructed to report various "environmental exposures."
1)A male participant who is receiving or has discontinued study intervention exposes a female partner prior to or around the time of conception."
2) "A female family member or healthcare provider reports that she is pregnant after having been exposed to the study intervention by inhalation or skin contact."
3) "A male family member or healthcare provider who has been exposed to the study intervention by inhalation or skin contact then exposes his female partner prior to or around the time of conception."
4) "A female is found to be breastfeeding while being exposed or having been exposed to study intervention (ie, environmental exposure). An example of environmental exposure during breastfeeding is a female family member or healthcare provider who reports that she is breastfeeding after having been exposed to the study intervention by inhalation or skin contact."trial who had close contact to a vaccinated person:
From Banoun:
The protocol for the Pfizer Phase I/II/III trial of COVID-19 mRNA vaccines (which began in May 2020) mentions the possibility of passage of the study product through inhalation or skin contact and passage through semen from a man exposed through inhalation or skin contact and passage through breast milk; the possibility of an adverse vaccine reaction from these exposures is also mentioned [15]. Pfizer's data clearly indicate that a pregnant woman may be exposed to “the intervention studied due to environmental exposure.”
Environmental exposure can occur through “inhalation or skin contact.” Examples of environmental exposure during pregnancy include: A female family member or health care provider reports that she is pregnant after being exposed to the study intervention through inhalation or skin contact. A male family member or health care provider who was exposed to the study intervention by inhalation or skin contact subsequently exposes his female partner before or around the time of conception.
Please re-read that last sentence again as I think it is critically important to understand what they are describing, i.e. “secondary shedding,” meaning someone can be “exposed” via inhalation or skin contact and then secondarily “expose” someone else. This will be important to remember in later posts where I provide clinical case examples of such “secondary shedding” events causing symptoms.
Banoun further interprets the section as follows:
This clearly means that any contact, including sexual contact with someone who has received the vaccines, exposes those who have not received the vaccines to the “intervention”, i.e. mRNA. Exposure during breastfeeding had also to be immediately notified during the trial: it is assumed that the investigator is concerned that a breastfeeding mother could transmit the experimental mRNA to her baby if she received the vaccines directly or if she is “exposed to the study intervention by inhalation or skin contact.”
Also remember the mention of sexual contact. Part 7 of this series describes two clinical reports of symptoms occurring immediately after a particular type of sexual intercourse (although a short post, for sensitive reasons I put it behind a paywall however numerous other clinical examples are provided in other posts in this series).
Unsurprisingly, numerous fact check articles were published to refute the above interpretations of the trial protocol language. This Chicago Tribune article argues that the language does not mean that Pfizer is suggesting that shedding can occur. To make this argument, they found this completely random, unknown professor who explained it away as follows:
Dr. Shobha Swaminathan, an associate professor of medicine at Rutgers New Jersey Medical School, referred to the document’s language as “generic” meant to cover cases of “any potential exposures, including possible accidental ones.”
Swaminathan said that “exposure” through inhalation or skin contact could refer to incidents where a pregnant woman was near a syringe of the product that accidentally broke. But in the case of COVID-19 vaccines, the degree of absorption from spilling the vaccine on your skin is “probably going to be negligible to non-existent,” Swaminathan said.
“Absorption will probably be negligible.” I am not reassured by Professor Swaminathan’s confidence. “In case a syringe accidentally broke.” I didn’t know that glass syringes were still in widespread use. The fact checkers are having a tough time here.
Sasha Latypova recently sent me another piece of evidence that Pharma knows that gene therapy product shedding occurs. The following is from an insert of a drug called Luxterna, an adenovirus vector gene therapy injected into the retina (IMO a terrible drug given that serious injuries impacting vision occurred in over 5% of subjects). Read on:
Links to all the other already active posts in this series is after the subscribe button below.
P.S. I just want to say thanks to all my subscribers, especially the paid ones! Your financial support is greatly appreciated as it allows me to devote what is often large amount of time I spend researching and writing my posts, so again, thanks. - Pierre
“Shedding” Part 1- Shedding of Covid mRNA Vaccine Components and Products From The Vaccinated to the Unvaccinated - Part 1
“Shedding” Part 2 - The Bio-Distribution and Excretion Potential of Covid mRNA Vaccine Products
“Shedding” Part 3 - Can You Absorb Lipid Nanoparticles From Being Exposed To a Vaccinated Person?
“Shedding” Part 4 - Evidence of Placental and Breast Milk Transmission of Covid mRNA Vaccine Components
"Shedding" Part 5 - Evidence of Shedding Causing Illness In Others
“Shedding Part 6- Clinical Case Notes Describing Shedding Phenomena Among Leading Edge Clinic Patients
“Shedding” Part 7 - Shedding Via Sexual Intercourse - Clinical Reports
“Shedding Part 8” - A Deluge of Clinical Reports Pour In
“Shedding” Part 9 - More and More Clinical Case Descriptions of Shedding Pour In
P.P.S - Proud to report that my book is sometimes gaining Best Seller status on Amazon in several countries and is climbing up the U.S Amazon rankings… Link:







Re “Shedding studies were not done because, even though the vaccines are gene-therapies, they legally fell under the legal definition of a “countermeasure” in a public health emergency.”
What exactly was the ‘public health emergency’?
There never was a ‘public health emergency’. It was a ‘PHEIC’ aka fake.
This fiasco has been a deliberately manufactured crisis to facilitate a massive vaccine market and control of the people.
"Dr. Shobha Swaminathan, an associate professor of medicine at Rutgers New Jersey Medical School, . . ."
I think that it's important to remember that Rutgers is intimately connected ($) to the Covid vaccine project and still has a vax mandate in place.