I Was Wrong About Structured Water
That realization led to something more scientifically accurate and useful: "cosmotropic water," a mineral-defined framework that now underpins a new category of agricultural input.
Dear Readers,
You and our Leading Edge Clinic patients make up the entire customer base of the Asao Group to this point. The broader world has yet to discover Themarox or Shimanishi, at least until the books are out, and in the meantime, you’ve kept us more than busy!
With that, I want to remind you that this weekend is our Spring Sale in celebration of World Water Day tomorrow: 25% off with code SPRINGSALE26 on single and paired bottles of Aurmina (drinking water) and Primora Bio (soil and foliar applications), plus additional discounts on a variety of bundles.
The Structured Water Question
After seven months chasing minerals through water chemistry, geohydrology, origin-of-life science, and old biophysics papers, I’ve arrived at an uncomfortable conclusion: I can’t prove that structured water exists, but I also can’t dismiss it. What follows is the evidence, so you can decide where you land.
I think the reader deserves a fair tour of why the structured-water idea refuses to die, why so many serious scientists have proposed it for over 100 years, and why I eventually had to leave that phrase behind without entirely leaving the question.
The Example of Ice
The safest place to begin is with something utterly uncontroversial: ice. No one in water chemistry can dispute that, under most conditions, when water freezes, its molecules arrange themselves into a highly ordered crystalline lattice, a stable, repeating architecture driven by polarity and hydrogen bonding. That matters because it establishes the first principle cleanly. Water is not some chemically indifferent fluid incapable of order. It carries within its molecular design the capacity to organize.
But ice does not solve the real question, and in some ways it distracts from it. Biology cannot run inside a rigid crystal as metabolism requires mobility, exchange, diffusion, slippage, collision, and all the molecular untidiness of liquid life.
Bulk Water: Organized Chaos
Imagine you could peer into a glass of pure liquid water and watch the individual molecules interacting with one another. What you would see would be astonishingly dynamic. Water molecules continuously form and break hydrogen bonds with neighboring molecules at the rate of hundreds of billions of times per second, and although they often adopt tetrahedral arrangements for brief moments, those structures are so short-lived that thermal motion tears them apart almost as quickly as they appear. Clusters form and dissolve almost instantly, and the best description of the whole scene is probably “organized chaos.”
That, more or less, is the baseline state of liquid water: a rapidly shifting hydrogen-bond network with fleeting local arrangements and no durable large-scale order. And if that were all water ever was, the structured-water question would never have become a question in the first place. But of course, that is not all water ever is.
Minerals Bringing Order To Water: Hydration Shells
OK, so I have to admit that I just played a trick on you. I had you imagine a glass containing only water molecules, which is to say a liquid that does not exist in nature outside laboratory conditions or with devices, such as ultrapure, distilled, or reverse-osmosis water. My bad. It is almost as if I believed that water without minerals is biologically active. It is not.
Add minerals back in, and the picture changes immediately. The moment a charged ion dissolves, nearby water molecules orient themselves around it, forming what chemists call hydration shells. I think of them as fans crowding around a celebrity—the closest press in tightly and most orderly, while those farther out remain influenced but more loosely arranged.
This is not a crystal and certainly not a frozen lattice, but it is a real pocket of local order created by mineral charge. The molecules remain in motion, the “fans” constantly exchanging places, but a general, albeit focal, structure persists through dynamic electrostatic relationships. When minerals enter the system, water does not stop being dynamic—it becomes dynamic around centers of organization. That is already more structure than one finds in demineralized water, though still far from the crystalline order of ice.
Water At The Border - Interfacial Water
The next level of order appears when water encounters a surface. Here, it becomes even more organized than in hydration shells, as electrostatic forces pull molecules into preferred orientations near charged or polar interfaces—membranes, mineral lattices, clays, proteins, while slowing their motion.
We are still on safe ground. Interfacial water is well established. Spectroscopic work confirms that water adjacent to mineral surfaces—such as those near my now-favorite pet rock, biotite—becomes more ordered than bulk water. The limitation, as water chemists insist, is that this ordering extends only a few nanometers, on the order of ten molecular layers.
Think of a Tokyo subway platform: the first row is highly organized, the second is still influenced, and farther out, you return to the chaotic movement of the crowd. That is interfacial water. The edge imposes order without freezing the system.
The Long Line of Structured Water Thinkers
Come with me now as we venture into the areas of “structured” water chemistry where assertions reach no consensus, and opinions on both sides get heated (you would think that water chemists are a boring bunch, but if you read the papers closely, it gets vicious (OK, OK, so I am over-injecting drama here, big deal).
But let’s start with the fact that far before the modern debates, for more than a century, scientists of the highest distinction, from very different fields, have asserted, in one language or another, that under certain conditions, the more organized behavior of interfacial water extends beyond the narrow layer recognized by classical surface chemistry.
Despite several leading, and in some cases, Nobel Prize–winning scientists proposing versions of the idea, to this day, no consensus exists on whether such states exist in the broader sense claimed or even on their biological importance. (Recall that “scientific consensus” is a trigger word for me).
If I were to give a full history of every study, lineage, and theoretical detour in this field, we would disappear into the weeds for far too long. So, let me try to keep it brief.
It all started with the Nobel Prize–winning biochemist Albert Szent-Györgyi, who suggested that water forms an active matrix within living systems, capable of supporting electronic and energetic organization around proteins.
It genuinely brought a smile to my face to come across Szent-Györgyi here. I had the same reaction months earlier when, during my dive into mineral science, I ran into two-time Nobel Prize winner Linus Pauling and that now-famous line often attributed to him:
“You can trace every sickness, every disease, and every ailment to a mineral deficiency.”
Why do I get a little thrill when names like Szent-Györgyi and Pauling show up in these corners of science?
My long-time readers can probably guess. My life—and, frankly, my career—collided with my dear friend and colleague Paul Marik through our shared fascination with the profound effects of IV vitamin C in severe sepsis. That path led us straight into COVID, and with it came… well, everything that followed—loss of jobs, careers, board certifications, and even our marriages.
What many of you may not know is that Szent-Györgyi won the Nobel Prize for discovering vitamin C, while Pauling was relentlessly criticized for promoting its therapeutic efficacy in cancer. Small world, right?
I continue to find it fascinating that the same names keep appearing in these seemingly distant corners of medicine and science that I keep wandering into. And no, before anyone gets carried away, I am not placing myself in the company of Nobel laureates, but, in a twist I still find surreal, I leave tomorrow for Japan for the first time in my life, where one of the highlights of the trip will be a personal invitation from Nobel Prize winner Professor Satoshi Ōmura, the discoverer of ivermectin, to tour the Kitasato Institute and share dinner with him and his colleagues.
I can tell you already, it will be one of the highlights of my life. I will never forget the kindness he showed me during Covid, when he reached out at a time I was at my lowest, sending a letter and a beautiful spectrograph of the bacteria Streptomyces Avermectin, a gift that, quite honestly, helped restore a level of strength and determination I might not have otherwise regained.
On top of that, I’ll be meeting with the executives of the Shimanishi-Kaken company and touring the only facility in the world that produces Themarox from biotite-derived vermiculite. Not a bad week.
Meanwhile, Lisa has been thoroughly entertained by watching me learn from YouTube videos about the three types of bows used in Japan. Apparently, the angle matters depending on who you’re greeting. If you wait until the end of this post, I will share the video she took of my lesson with her. But suffice it to say, I’ve also been consulting AI extensively on Japanese dining etiquette and conversation customs.
Because anyone who knows me knows this: I require serious training if I’m going to avoid playing the loud, overly boisterous American.
Anyway, where was I? Oh yeah, a few decades after Szent-Györgyi, the biophysicist Gilbert Ling proposed that intracellular water exists in a more ordered state associated with protein surfaces, formalizing the idea in his association–induction hypothesis. Around the same time, the surface chemist Boris Derjaguin demonstrated experimentally that water adjacent to solid materials, including mica and other mineral lattices, forms structured interfacial layers with properties distinct from bulk liquid water.
Later, the Italian physicist Emilio Del Giudice introduced a theoretical framework suggesting that water might organize into coherent domains through electromagnetic coupling. Taken together, these scientists were not saying exactly the same thing, but they were all, in their own language, pushing against the idea that liquid water should always be treated as a simple, chaotic, structureless medium.
Enter Gerald Pollack
Which brings us to the work of Professor Gerald Pollack at the University of Washington. Pollack credits his interest in the subject to reading Gilbert Ling, and his exclusion-zone experiments can be understood as a modern extension of Ling’s long-running line of inquiry into whether water in biological systems behaves differently from bulk liquid.
What Pollack reported was the discovery of relatively large regions of water adjacent to hydrophilic surfaces in which particles and solutes were excluded, an observation that implied that the water molecules within that region had adopted a more ordered arrangement than the surrounding bulk liquid. In other words, something about proximity to a surface appeared to reorganize the water in a way that fundamentally changed its behavior.
These regions, which he called “exclusion zones,” were not just small boundary effects. He reported that they extended roughly 10,000 to 100,000 times farther than the nanometer-scale interfacial layers traditionally described by surface chemistry. That claim alone was enough to turn heads, and, predictably, to attract a small but enthusiastic horde of detractors within the water chemistry community. Science, after all, has its own immune system, and it tends to react when something new shows up that it didn’t generate itself.
Pollack’s experiments were also visually striking, which matters more than most scientists would like to admit. He was able to literally show these zones forming and expanding under the microscope. Because he named the phenomenon in a way that people could understand, the idea began to spread beyond the lab. He reported that EZ water carried a net negative charge, while the adjacent bulk water carried a positive charge, effectively creating a region of charge separation, a kind of stored potential energy within the system.
Even more intriguing, when infrared light was applied, the exclusion zone expanded. Others observed that healthier cells appeared to contain more of this EZ-like water, while damaged or dying cells contained less, and Pollack himself reported that certain toxins caused the zone to shrink. The implication, never stated too bluntly, but always hovering nearby, was that the amount of this organized water might correlate with biological health.
From there, Pollack made a bold interpretive leap. He proposed that these regions represented a distinct, previously unrecognized phase of water, a “fourth phase,” somewhere between liquid and solid, almost gel-like, in which water molecules were organized into stacked sheets forming a repeating lattice. He titled his book The Fourth Phase of Water, and from that moment on, the language took on a life of its own.
And, as tends to happen, it didn’t stay confined to the laboratory. The concept migrated, first into broader scientific curiosity, then into wellness culture, then into device marketing, and finally into all the familiar corners where incomplete science becomes a commercial opportunity. You can probably guess the rest.
One of the most important experimental findings in Pollack’s work was that EZ water could be identified by a characteristic ultraviolet absorption peak at around 270 nanometers. That was his proposed signature. Fine. But then, in interviews and public talks, he began to mention that similar spectral features could be found in certain natural waters, particularly pristine mountain spring waters, with some showing more pronounced peaks than others.
Now, whether intentional or not, that suggestion carried implications. It subtly suggested that some waters might be inherently more “organized” and therefore more biologically beneficial than others. Predictably, that idea didn’t stay theoretical for long. An entire industry of devices emerged, claiming to “structure” water using magnets, fields, vortices, or various energetic inputs, often without, I should note, providing clear evidence that their treated water actually exhibited the very 270 nm signature Pollack had identified as central. That detail, as you will see later, becomes quite important.
Stepping back, I found Pollack’s body of work genuinely compelling. His findings around exclusion, charge separation, infrared responsiveness, and potential biological relevance were not hand-wavy, they were observed, tested, and published. One of the more striking claims was that this charge separation could even contribute to capillary flow independent of cardiac pumping, with experiments suggesting sustained movement under certain conditions. That is not a trivial observation.
After reviewing his work and learning about a conversation my colleague Matt Bakos had with him, I have no issue with the phenomenon he described. Where my questions began was not with what he observed, but with the conditions under which those observations were made, and whether those conditions reflected the way water actually exists in the real world.
The Opposition
Pollack’s critics, to be fair, have offered many alternative explanations for exclusion-zone behavior, attributing it to well-understood concepts such as diffusion gradients, surface charge effects, diffusiophoresis, and the specific chemistry of the materials used.
I take those seriously. But what left me unconvinced was the lack of a single elegant counter-explanation that accounted for the whole set of observations in a way that was more coherent than Pollack’s own. Different rebuttals seemed to be deployed for different pieces of the phenomenon.
That does not vindicate Pollack’s strongest claims, and I do not need it to. It merely suggests that interfacial water, mineralized water, and electrochemical surface behavior remain more interesting than the dismissive crowd would like them to be.
Ultimately, I think of Pollack’s novel insights like any other in science, they pass through three stages: first, they are observed, then they are argued over, and only later—if they survive—are they explained well enough to be accepted. The truth is that Pollack’s ideas, at the time of this writing, are still stuck in the second stage.
Where Shimanishi Convinced Me Even Further
Despite that, after wading through all the controversy and debate in really dense water chemistry papers, I was still left with the impression that something real was being observed. Still, the interpretation remained so unsettled that I decided to stop making any more assertions about structured water.
But then, as I was about to leave the subject for good, I was drawn back in when I noticed uncanny parallels between Shimanishi's numerous claims and findings, which not only predated Pollack by a decade but also almost completely mirrored them.
I also realized an important distinction: Shimanshi’s observations did not arise from working with synthetic Nafion tubes and injected microbubbles; he was working in the real world, with real water, not lab water.
Despite this difference, the more I looked, the more intriguing overlaps I noticed. Pollack maintained that exclusion-zone water showed a characteristic UV absorption peak around 270 nanometers. Themarox-treated water, when tested both in Pollack’s laboratory and independently, showed the same peak.
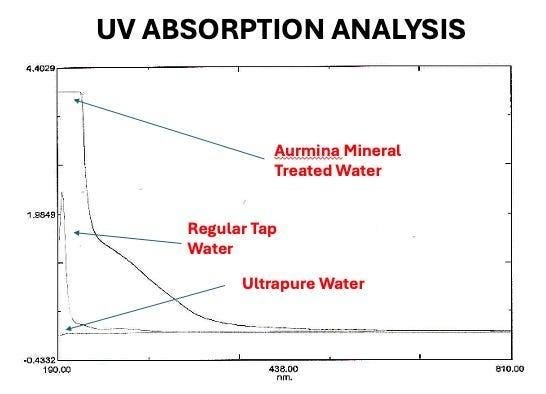
UV Spectrometer Analysis Of Water Treated With A Themarox-Derived Solution
In the below, the absorption of Themarox-treated water was compared to both regular tap water and “Ultrapure” water (a completely de-ionized (zero mineral content) water.
A critical detail is that the treated water sat in the lab for 6 days prior to the test.
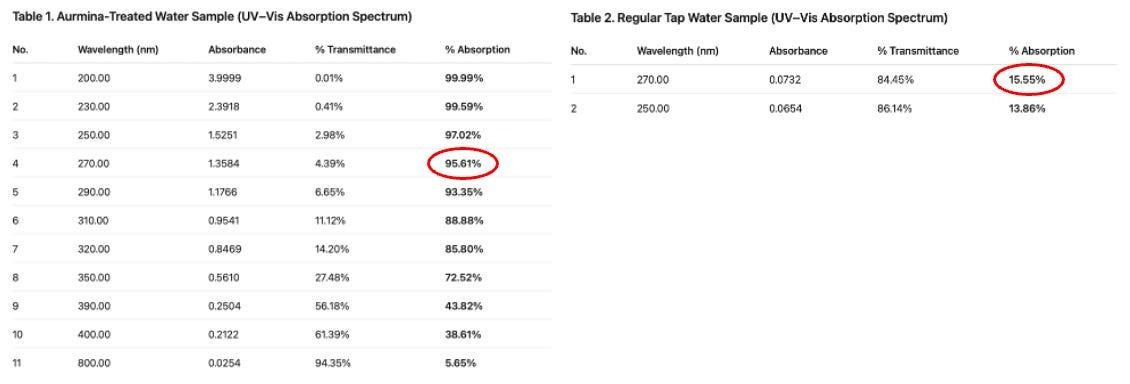
In the above, you can see the long “hold” where UV absorption is occurring in the 6-day-old mineral-treated water. In the tables below, note the sulfated mineral-treated water showing 95% absorption at 270 nm compared to the 15% absorption in tap water, and 0% in Ultrapure water:
The coincidence was too specific to dismiss casually.
Then the parallels widened. Pollack claimed that EZ water excluded suspended particles and solutes from the organized region; Themarox-treated water, when added to ordinary modern water, rapidly promotes aggregation, separation, and settling of suspended material, leaving the overlying water visibly clearer. The conventional explanation is easy; classical flocculation, which may be entirely sufficient to account for the result, as it is well known that multivalent ions destabilize colloids.
But what caught my attention was that Shimanishi, long before Pollack, described his treated water as reorganized into smaller water clusters of H₃O₂, exactly like Pollack described EZ water clusters, and Shimanishi also claimed that, based on this organization, it further allowed his treated water to expel solutes and particles from solution.
Shimanishi supported his claim not with UV absorption but with results from nuclear magnetic resonance (NMR) testing, which he interpreted as representing unique hydrogen-bond dynamics.
Weird right?
Shimanishi arrives at this conclusion by himself, using “real,”, “natural”, mineral-treated water samples and then, 10-15 years later, a guy named Pollack in a research lab at the University of Washington, arrives at the same conclusion, mechanism, and assertions, except that, instead of using NMR, he suggests that a 270um UV absorption peak is the identifier of such water structure.
It gets worse (or better). The electrochemistry of the different water samples offered yet another point of overlap. Pollack described charge separation and potential differences in exclusion-zone systems. Themarox-treated water also showed measurable electrochemical shifts, including changes in redox chemistry, oxygen demand, colloid stability, and microbial burden.
But again, the “consensus” among water chemists argues for more conventional explanations for this phenomenon: hydrolysis of iron and aluminum species, double-layer compression, flocculation, and oxidation of dissolved organics. Fine. But once enough of these overlaps accumulate, I find it difficult not to ask whether two apparently different systems may be arriving at a shared class of behavior through different routes.
The Missing Variable: Mineral Composition
That was the point at which I began to suspect that the real missing variable in the entire structured-water conversation was minerals.
This is a critical point: Pollack’s experiments were done in demineralized water, which makes perfect laboratory sense if one is trying to isolate surface effects. But every other earlier scientist, including Shimanishi, who was interested in biologically active water, studied water as it actually exists in life and geology: mineralized, ionic, embedded in gradients, and interacting with charged surfaces.
Pollack’s system, by contrast, stripped all of that chemistry away. Which, in one sense, was fantastic. Pollack was able to create water with similar properties under controlled conditions, allowing him to describe its properties and behaviors in ways not possible in nature!
Still, to my mind, that is not a trivial difference. Water in biology is never just H₂O. In geology, water is never just H₂O. Water in a cell, around a protein, inside a soil pore, along a clay sheet, or moving through a fractured mineral system is always a mineral solution. Its conductivity, buffering behavior, redox activity, interfacial dynamics, and biological compatibility all arise from that composition. Remove the minerals, and you have not created natural water; you have created an experimental medium. Useful for conducting precise investigations, yes. Biologically representative, no.
Once that realization settled in, the structured-water question shifted for me. I found myself more interested in the ionic environments it carries, the surfaces it touches, and how those variables reorganize the medium.
In that light, Pollack’s use of synthetic Nafion tubes became newly suggestive. Nafion is a synthetic polymer, yes, but it behaves in some respects like an intensely charged mineral surface. Its fixed negative charges mimic a crucial feature of natural mineral interfaces. Which means that Pollack’s experimental system may have been reproducing, in a stripped-down synthetic form, something that water encounters continuously in geological environments.
That possibility pulled me back toward Shimanishi. Themarox does not merely “purify” water. It introduces a specific mineral spectrum, one rich in strongly “cosmotropic” ions derived from a weathered vermiculite lattice. Once dissolved, those ions alter the ionic field, change interfacial behavior, reshape electrochemical conditions, and create an environment in which water no longer behaves like a generic bulk fluid.
Seen in that light, the overlap between Pollack’s findings and Shimanishi’s observations begins to look less like a coincidence and more like two different experimental paths stumbling toward the same chemical territory.
The Structured Water Device Industry
At that point, I also began to see more clearly why the structured-water device industry had gone off the rails. Once you remove minerals from the center of the discussion, you create a vacuum that devices are only too happy to fill. Magnets, electrodes, vortices, frequency gadgets, and every other piece of expensive plumbing theater rush in to promise order from outside the system.
I do not deny that externally applied energy can transiently order or align water molecules. Thermodynamic principles have demonstrated that already. A whirlpool maintains its structure only while energy is moving through it. A hurricane exists only while its gradients persist. Cells, soils, and organisms are no different; they preserve order only so long as they can maintain gradients.
Which means that if one really wants to understand durable biological order, the question is not whether one can briefly perturb or organize water with some clever device. The question is whether the ionic and mineral framework of the medium can support stable electrochemical organization over time.
So that is where I now leave the structured-water question. Not solved. Not embraced as a slogan. Not discarded. Narrowed. I no longer need to insist that Shimanishi’s mineral system “structures” water in the way the popular term implies. What I need, and what the next step in the argument gives me, is something older, firmer, and much more precise.
Once mineral surfaces are taken into account, dissolved ions cannot be treated as background. Natural waters are mineral solutions, and the ions they carry systematically alter hydration, protein stability, interfacial charge, and molecular organization.
Franz Hofmeister recognized this more than a century ago. And once I finally let that sink in, the entire conversation became far more powerful, because we were no longer arguing over slogans. We were back on the solid ground of chemistry.
Hoffmeister described a series of mineral-specific effects, showing how different minerals influence protein stability, surface tension, interfacial charge, and molecular organization throughout biological systems. Put simply, the structure and behavior of water are determined by the minerals dissolved within it.
And that, for my purposes, is where the discussion becomes both older and more precise. Because once the conversation shifts from sweeping claims about “structured water” to the specific and measurable effects of dissolved ions on hydration shells, interfaces, proteins, and gradients, we leave the land of slogans and enter the much firmer terrain of Hofmeister chemistry.
That realization is what led directly to my insight into Primora Bio as a deliberately constructed mineral environment, one designed by nature to shift water toward a more stable, cosmotropic state, where organization is not imposed from the outside, but emerges from the chemistry itself.
And once you see water that way, it becomes very difficult to see it any other way again.
*If you value the late nights and deep dives into all the “rabbit holes” I write about (or the Op-Eds and lectures I generate for the public), your support is greatly appreciated.
Bonus fun: Video of me teaching Lisa the various Japanese bows we need to use next week:
World Water Day Sale - Discount Code: Springsale26
Sale: 25% off all single bottles of Aurmina or Primora Bio, with separate, already-included bundle discounts ranging from 25% to 30% on multi-bottle and combination bundles.
UK, European Customers: Our first UK distributor, Nicholas Smith at The Water Dr., is offering a 10% discount until the end of the month, with coupon code KORYUK10. Much larger savings will be made by reducing shipping costs, given his 1-bottle fixed-price delivery of £5 for the UK and £15 for the EU (standard tracked and insured).
Regenerative Farming Starter Kit: For readers looking forward to kickstarting their best summer garden ever, spring planting is around the corner, and I am most excited about our “Regenerative Farming Starter Kit,” which includes Primora Bio (cosmotropic water concentrate), Primora Char (high-grade liquid biochar), and Primora Nourish (liquid fish hydrolysate fertilizer).
*I have not written about the wonders of biochar or fish hydrolysate, but will soon.





Interesting you mentioned ice. As a pastor for many years, I have tried to wrap my mind around the nature of God's Personhood. He is Trinity - Father, Son, Spirit. According to the understandings of early church counsels (and Scripture) each person is of the same essence - fully God, but not three Gods. My best understanding is this: water, which exists in 3 forms - ice, liquid and vapor. But they do not exist so simultaneously. If water could be ice, liquid and vapor all at the same time, one might have a little, human idea of Him. Just thinking.
Refreshing seeing a medical professional say they were wrong.